Decision-making process
Information box
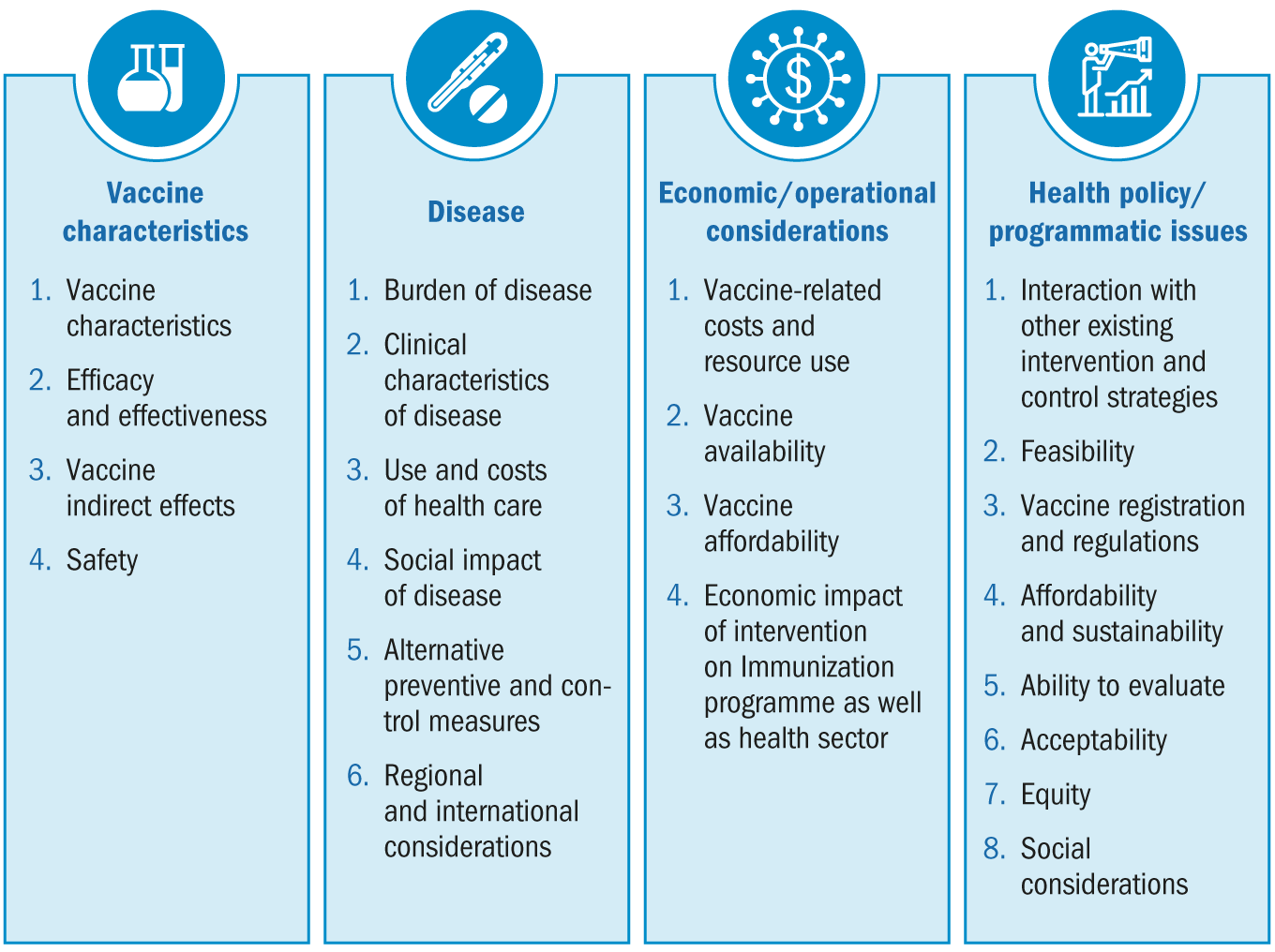
As with other vaccines, the Ministry of Health should request the NITAG to conduct a review of local and global evidence. This includes disease characteristics (e.g. influenza disease burden among pregnant women and children under 6 months of age, influenza virus seasonality and antigenicity), vaccines and immunization characteristics (e.g. effectiveness and safety characteristics of available vaccines), economic and operational considerations (e.g. vaccine availability, affordability, vaccine costs and resource use, economic impact), and health policy and programmatic issues (e.g. interaction with other interventions, feasibility, acceptability) (see figure below). Where available, the NITAGs may draw some of the information required from National influenza centres that are tasked with monitoring influenza virus composition in a number of countries.
A review of existing vaccination activities (e.g. maternal tetanus toxoid or other vaccination efforts targeting pregnant women) may provide useful information on delivery of vaccine via the antenatal care system. Such a review of existing delivery strategies should include coverage rates, vaccine acceptability and any logistical challenges identified.
NITAGs or equivalent technical advisory bodies can provide transparent and independent recommendations to the Ministry of Health. Additional information should be considered by the Ministry of Health from other relevant groups of which can be either members of NITAGs (core, ex-officio, or liason members) or should be involved in development of NITAGs recommendations through participation in working groups.
These can include other ministries (e.g. Ministry of Education, Ministry of Defence, Ministry of Finance) as well as from academic, scientific and professional groups (e.g. antenatal care providers such as obstetricians, midwives and family or general practice physicians), professional associations (e.g. associations of gynaecologists, paediatricians), civil society organizations, the private sector, and high-level national stakeholders that could champion and disseminate information on maternal influenza vaccination. In particular, the inclusion of professional organizations (e.g. general practitioners, gynaecologists, midwives) is relevant to ensure agreement with national recommendations and professional buy-in during the further roll-out of the programme.
The objectivity and independence of NITAG members will enhance the group’s credibility and strengthen the argument for securing funding from national authorities or donors for the introduction of the vaccine based on an evidence-based recommendation.
Elements that should be assessed, discussed and addressed by NITAGS during the introduction of maternal influenza vaccination
On the basis of the evidence reviewed, the EPI manager and the programme manager for reproductive, maternal, newborn, child and adolescent health (RMNCAH) or equivalent should provide recommendations on the planning of vaccine introduction to the Minister of Health who ultimately takes the decision to introduce the vaccine. Where available, an Interagency Coordinating Committee (ICC) or equivalent body, made up of representatives of the Ministry of Health, WHO, UNICEF, and other domestic and external partners, should, as a multidisciplinary group, help to guarantee coordination of implementation among partners and ensure funding within the national agenda for implementing influenza vaccination.
